2014
C. McGann, N. Akoum, A. Patel, E. Kholmovski, P. Revelo, K. Damal, B. Wilson, J. Cates, A. Harrison, R. Ranjan, N.S. Burgon, T. Greene, D. Kim, E.V. Dibella, D. Parker, R.S. MacLeod, N.F. Marrouche.
“Atrial fibrillation ablation outcome is predicted by left atrial remodeling on MRI,” In Circ Arrhythm Electrophysiol, Vol. 7, No. 1, pp. 23--30. 2014.
DOI: 10.1161/CIRCEP.113.000689
PubMed ID: 24363354
BACKGROUND:
Although catheter ablation therapy for atrial fibrillation (AF) is becoming more common, results vary widely, and patient selection criteria remain poorly defined. We hypothesized that late gadolinium enhancement MRI (LGE-MRI) can identify left atrial (LA) wall structural remodeling (SRM) and stratify patients who are likely or not to benefit from ablation therapy.
2013
C. McGann, N. Akoum, A. Patel, E. Kholmovski, P. Revelo, K. Damal, B. Wilson, J. Cates, A. Harrison, R. Ranjan, N.S. Burgon, T. Greene, D. Kim, E.V.R. DiBella, D. Parker, R.S. MacLeod, N.F. Marrouche.
“Atrial Fibrillation Ablation Outcome is Predicted by Left Atrial Remodeling on MRI,” In Circulation: Arrhythmia and Electrophysiology, Note: Published online before print., December, 2013.
DOI: 10.1161/CIRCEP.113.000689
Background: While catheter ablation therapy for atrial fibrillation (AF) is becoming more common, results vary widely and patient selection criteria remain poorly defined. We hypothesized that late gadolinium enhancement magnetic resonance imaging (LGE-MRI) can identify left atrial (LA) wall structural remodeling (SRM) and stratify patients who are likely or not to benefit from ablation therapy.
Methods and Results: LGE-MRI was performed on 426 consecutive AF patients without contraindications to MRI and before undergoing their first ablation procedure and on 21 non-AF control subjects. Patients were categorized by SRM stage (I-IV) based on percentage of LA wall enhancement for correlation with procedure outcomes. Histological validation of SRM was performed comparing LGE-MRI to surgical biopsy. A total of 386 patients (91%) with adequate LGE-MRI scans were included in the study. Post-ablation, 123 (31.9%) experienced recurrent atrial arrhythmias over one-year follow-up. Recurrent arrhythmias (failed ablations) occurred at higher SRM stages with 28/133 (21.0%) stage I, 40/140 (29.3%) stage II, 24/71 (33.8%) stage III, and 30/42 (71.4%) stage IV. In multi-variate analysis, ablation outcome was best predicted by advanced SRM stage (hazard ratio (HR) 4.89; pKeywords: atrial fibrillation arrhythmia, catheter ablation, magnetic resonance imaging, remodeling, outcome
2011
C.J. McGann, E.G. Kholmovski, J.J. Blauer, S. Vijayakumar, T.S. Haslam, J.E. Cates, E.V. DiBella, N.S. Burgon, B. Wilson, A.J. Alexander, M.W. Prastawa, M. Daccarett, G. Vergara, N.W. Akoum, D.L. Parker, R.S. MacLeod, N.F. Marrouche.
“Dark Regions of No-Reflow on Late Gadolinium Enhancement Magnetic Resonance Imaging Result in Scar Formation After Atrial Fibrillation Ablation,” In Journal of the American College of Cardiology, Vol. 58, No. 2, pp. 177--185. 2011.
DOI: 10.1016/j.jacc.2011.04.008
PubMed ID: 21718914
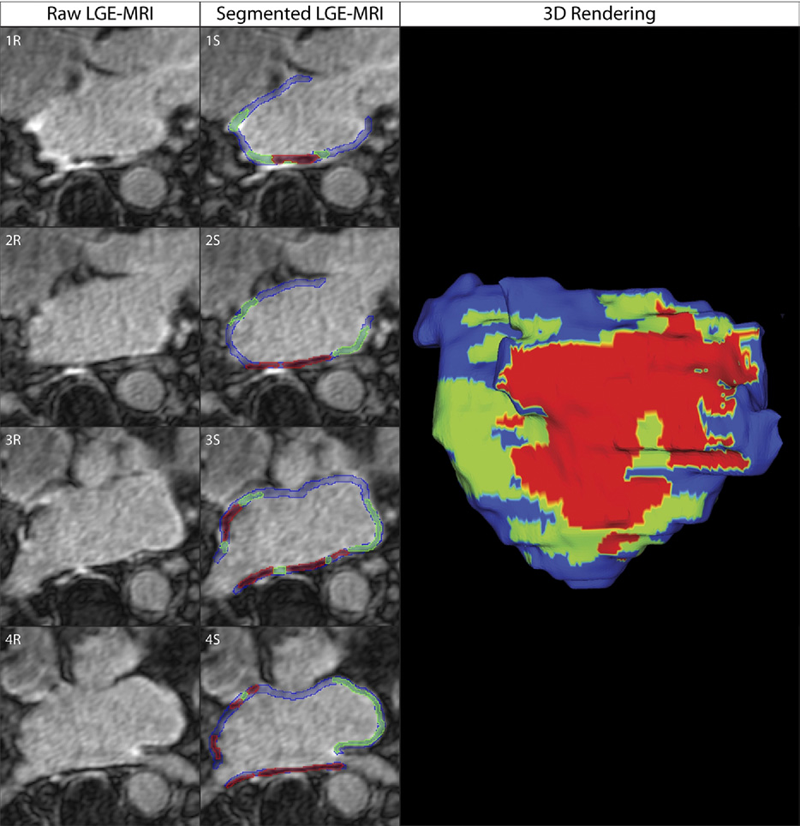
Objectives: The aim of this study was to assess acute ablation injuries seen on late gadolinium enhancement (LGE) magnetic resonance imaging (MRI) immediately post-ablation (IPA) and the association with permanent scar 3 months post-ablation (3moPA).
Background: Success rates for atrial fibrillation catheter ablation vary significantly, in part because of limited information about the location, extent, and permanence of ablation injury at the time of procedure. Although the amount of scar on LGE MRI months after ablation correlates with procedure outcomes, early imaging predictors of scar remain elusive.
Methods: Thirty-seven patients presenting for atrial fibrillation ablation underwent high-resolution MRI with a 3-dimensional LGE sequence before ablation, IPA, and 3moPA using a 3-T scanner. The acute left atrial wall injuries on IPA scans were categorized as hyperenhancing (HE) or nonenhancing (NE) and compared with scar 3moPA.
Results: Heterogeneous injuries with HE and NE regions were identified in all patients. Dark NE regions in the left atrial wall on LGE MRI demonstrate findings similar to the \"no-reflow\" phenomenon. Although the left atrial wall showed similar amounts of HE, NE, and normal tissue IPA (37.7 ± 13\%, 34.3 ± 14\%, and 28.0 ± 11\%, respectively; p = NS), registration of IPA injuries with 3moPA scarring demonstrated that 59.0 ± 19\% of scar resulted from NE tissue, 30.6 ± 15\% from HE tissue, and 10.4 ± 5\% from tissue identified as normal. Paired t-test comparisons were all statistically significant among NE, HE, and normal tissue types (p less than 0.001). Arrhythmia recurrence at 1-year follow-up correlated with the degree of wall enhancement 3moPA (p = 0.02).
Conclusion: Radiofrequency ablation results in heterogeneous injury on LGE MRI with both HE and NE wall lesions. The NE lesions demonstrate no-reflow characteristics and reveal a better predictor of final scar at 3 months. Scar correlates with procedure outcomes, further highlighting the importance of early scar prediction. (J Am Coll Cardiol 2011;58:177–85) © 2011 by the American College of Cardiology Foundation
2010
S.K. Iyer, E. DiBella, T. Tasdizen.
“Edge enhanced spatio-temporal constrained reconstruction of undersampled dynamic contrast enhanced radial MRI,” In IEEE International Symposium on Biomedical Imaging (ISBI): From Nano to Macro, pp. 704--707. 2010.
DOI: 10.1109/ISBI.2010.5490077
S.S. Kuppahally, N. Akoum, N.S. Burgon, T.J. Badger, E.G. Kholmovski, S. Vijayakumar, S.N. Rao, J. Blauer, E.N. Fish, E.V. Dibella, R.S. Macleod, C. McGann, S.E. Litwin, N.F. Marrouche.
“Left atrial strain and strain rate in patients with paroxysmal and persistent atrial fibrillation: relationship to left atrial structural remodeling detected by delayed-enhancement MRI,” In Circ Cardiovasc Imaging, Vol. 3, No. 3, pp. 231--239. 2010.
PubMed ID: 20133512
N.M. Segerson, M. Daccarett, T.J. Badger, A. Shabaan, N. Akoum, E.N. Fish, S. Rao, N.S. Burgon, Y. Adjei-Poku, E. Kholmovski, S. Vijayakumar, E.V. DiBella, R.S. MacLeod, N.F. Marrouche.
“Magnetic resonance imaging-confirmed ablative debulking of the left atrial posterior wall and septum for treatment of persistent atrial fibrillation: rationale and initial experience,” In Journal of Cardiovascular Electrophysiology, Vol. 21, No. 2, pp. 126--132. 2010.
PubMed ID: 19804549
2009
R.S. Oakes, T.J. Badger, E.G. Kholmovski, N. Akoum, N.S. Burgon, E.N. Fish, J.J. Blauer, S.N. Rao, E.V. DiBella, N.M. Segerson, M. Daccarett, J. Windfelder, C.J. McGann, D.L. Parker, R.S. MacLeod, N.F. Marrouche.
“Detection and quantification of left atrial structural remodeling with delayed-enhancement magnetic resonance imaging in patients with atrial fibrillation,” In Circulation, Vol. 119, No. 13, pp. 1758--1767. 2009.
N.M. Segerson, M. Daccarett, T.J. Badger, A. Shabaan, N. Akoum, E.N. Fish, S. Rao, N.S. Burgon, Y. Adjei-Poku, E.G. Kholmovski, S. Vijayakumar, E.V.R. Dibella, R.S. Macleod, N.F. Marrouche.
“Magnetic Resonance Imaging-Confirmed Ablative Debulking of the Left Atrial Posterior Wall and Septum for Treatment of Persistent Atrial Fibrillation: Rationale and Initial Experience,” In Journal of Cardiovascular Electrophysiology, Vol. 21, No. 2, pp. 126--132. 2009.
2008
C.J. McGann, E.G. Kholmovski, R.S. Oakes, J.J. Blauer, M. Daccarett, N. Segerson, K.J. Airey, N. Akoum, E. Fish, T.J. Badger, E.V. DiBella, D.L. Parker, R.S. MacLeod, N.F. Marrouche.
“New Magnetic Resonance Imaging-Based Method for Defining the Extent of Left Atrial Wall Injury After the Ablation of Atrial Fibrillation,” In Journal of the American College of Cardiology, Vol. 52, No. 15, pp. 1263--1271. Oct 7, 2008.
2007
G. Adluru, S.P. Awate, T. Tasdizen, R.T. Whitaker, E.V.R. DiBella.
“Temporally Constrained Reconstruction of Dynamic Cardiac Perfusion MRI,” In Magnetic Resonance in Medicine, Vol. 57, pp. 1027--1036. 2007.
G. Adluru, R.T. Whitaker, E.V.R. DiBella.
“Spatio-Temporal Constrained Reconstruction of Sparse Dynamic Contrast Enhanced Radial MRI Data,” In Proceedings of the IEEE International Symposium on Biomedical Imaging, pp. 109--112. 2007.
G. Adluru, E. Hsu, E.V.R. DiBella.
“Constrained Reconstruction of Sparse Cardiac MR DTI Data,” In Proceedings of FIMH 2007, LNCS, No. 4466, pp. 91--99. 2007.
2006
G. Adluru, E.V.R. DiBella, R.T. Whitaker.
“Automatic Segmentation of Cardiac Short Axis Slices in Perfusion MRI,” In Proceedings of The 2006 IEEE International Symposium on Biomedical Imaging, pp. 133--136. 2006.
G. Adluru, E.V.R. DiBella.
“Segmentation Based Registration of Myocardium in Cardiac Perfusion Images,” In Proceedings of The 14th Annual Scientific Meeting of The International Society for Magnetic Resonance in Medicine (ISMRM), Vol. 14, pp. 1223. 2006.
L. Lorenzo, R.S. MacLeod, R.T. Whitaker, G. Adluru, E.V.R. DiBella.
“Level Sets and Shape Models for Segmentation of Cardiac Perfusion MRI,” In Proceedings of International Society for Optical Engineering (SPIE) Conference on Medical Imaging 2006, Vol. 6144, pp. 1320--1331. 2006.
2005
E.G. Khölmovski, E.V.R. DiBella, A.A. Samsonov.
“Accurate Assessment of the Arterial Input Function using MRI with Radial Sampling,” In Proceedings of the Society for Cardiovascular Magnetic Resonance (SCMR) 8th Annual Scientific Sessions, Vol. 7, pp. 292--294. 2005.
A.A. Samsonov, E.V.R. DiBella, P. Kellman, E.G. Kholmovski, C.R. Johnson.
“Adaptive k-t BLAST/k-t SENSE for Accelerating Cardiac Perfusion MRI,” In Proceedings of the Society for Cardiovascular Magnetic Resonance (SCMR) 8th Annual Scientific Sessions, pp. 277--278. 2005.
2002
R.S. MacLeod, E.D. DiBella, B.B. Punske, E.K. Jeong.
“Multimodal Imaging of Cardiac Activation, Perfusion, and Ischemia,” In 14th Annual Research Symposium of the Medical Imaging Research Laboratory, 2002.